History of OxyContin
Purdue created OxyContin after the global medical community expressed grave concern about the undertreatment of pain. It is a long-lasting version of the commonly prescribed generic medication oxycodone, which was first introduced in 1916.
FDA has reviewed OxyContin multiple times over the past 25 years – by different review teams under different political administrations – and has repeatedly determined it is safe and effective when used properly.
OxyContin can only be obtained lawfully through a DEA-registered physician’s prescription. The doctor, in consultation with the patient, determines the appropriateness of opioids for that patient’s specific circumstances, taking into consideration the known risks of addiction. Importantly, Purdue is not involved with this decision.
Properly prescribed opioids are essential to the treatment of chronic severe pain and offer relief to millions of people. The risk of abuse, addiction, and death has always been prominently disclosed in OxyContin’s original and subsequent labels.
OxyContin: One Part in Long History of Prescription Opioids
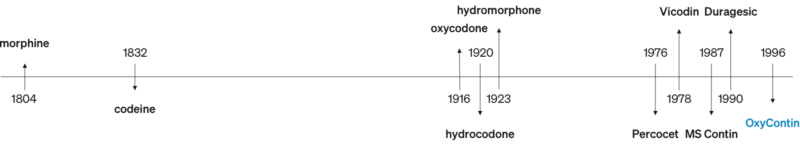
OxyContin was introduced in 1996, following a long line of prescription opioids being developed to treat pain.
OxyContin Introduced as First Extended-Release Form of oxycodone
OxyContin was developed as an extended-release version of the generic, short-acting oxycodone medicine originally introduced in 1916. Patients who are prescribed extended-release medicines by a physician are able to take fewer doses throughout the day and therefore experience less sleep disturbance
Extended-release versions of other generic medicines had been prescribed by physicians as early as 1947 when Dolophine was introduced.
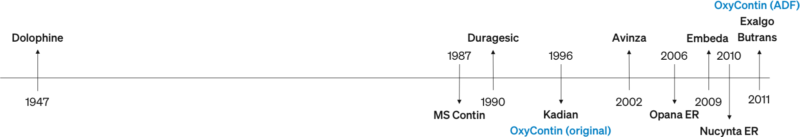
Competitive Landscape of Extended-Release Opioids
Prescription Opioid Overview
Risk of Addiction Always Disclosed
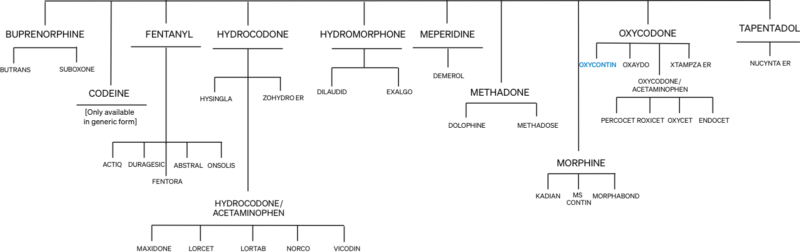
All opioid medicines have always been known to carry risk of addiction and abuse, which is why they are Schedule II medications. The risk of abuse, addiction, and death has always been prominently disclosed in OxyContin’s original and subsequent labels.
Neither the FDA nor Purdue Pharma anticipated the level of abuse that occurred. In fact, they expected any abuse of OxyContin would be similar to the experience with Purdue’s long-acting morphine medication, MS Contin (introduced in 1987).
National Media Coverage of OxyContin Misuse Emerged in Late 2000 & 2001, Following News Reports About Other Opioid Medications
Purdue then took prompt action when the problem became understood.
Drug Use Weakened Allegations of Abuse
“Vicodin is abused a lot,” said Robert Popovian, a doctor of pharmacy and a researcher at USC.
SEPTEMBER 10, 1995[1]
“It’s a problem.”
Vicodin, My Vicodin
“It is the painkiller of choice for thousands of drug users, legal and illegal both.”
JANUARY 1, 1999[2]
Misuse of Pain Drug Linked to Hearing Loss
“Vicodin, one of the most commonly prescribed painkillers, is frequently used improperly.”
SEPTEMBER 10, 2001[3]
Some of This Media Coverage Included Specific Directions on How to Use OxyContin Improperly for Recreational Purposes
The Potent Perils of a Miracle Drug
“In any given week, her husband reportedly told investigators, the couple supplemented their Social Security by selling drug addicts $8,000 worth of the tiny white tablets that are chewed or smashed to remove the time-release coating, then snorted or injected, generating a high as intoxicating as that of heroin.”
TIMOTHY ROCHE
DECEMBER 31, 2000[4]
The Alchemy of OxyContin
“But these days, she says, the only drug for sale in Man is OxyContin, a narcotic painkiller that users crush — to disable its patented time-release mechanism — and then snort or inject for a powerful and immediate opiate high. Legally, it’s sold only by prescription for the treatment of chronic pain. In practice it’s available just about everywhere around here, immediately, for cash. The going rate is a dollar a milligram, or $40 for a 40-milligram pill.”
PAUL TOUGH
JULY 29, 2001[5]
Doctors & Patients Continue to Express Need for OxyContin
In April 2020, CDC and HHS requested comments “concerning perspectives on and experiences with pain and pain management” to help update the 2016 guidelines on opioid prescribing.[6]
“I am only able to function daily because of the Oxycontin Extended Release.”[7]
“… I have been on oxycontin ER for 14 years and it has been life changing for the better. When the CDC changed the guidelines in 2016 my Dr was threatened and he had to taper me down to the 90mm suggested by the CDC. For over 3 years my life has deteriorated and my pain is horrible…”[8]
“…taking this opioid drug allowed me to control and manage that pain so I could have some quality of life.”[9]
“…Oxycontin has saved my life!!”[10]
“I will advocate for the continued legalization and availability of opiate pain medications, including Norco, OxyContin, Dilaudid, Morphine, and Fentanyl Patches, when needed to mitigate chronic pain for pain sufferers like me.”[11]